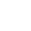Leader nella produzione di sale marino pregiato e totalmente naturale.
Salute, ambiente, innovazione, qualità: sono questi i valori che fanno di CIS un’azienda sana.
Scopri CIS



IL FRUTTO DEI
NOSTRI MARI
Tutte le forme del nostro sale
COMPAGNIA ITALIANA SALI
Il nostro processo di produzione e supply chain va dall'estrazione alla distribuzione.
Scopri di più
Un’ampia gamma, forte di diversi marchi che consentono di soddisfare tutte le esigenze dei consumatori.













Il Fior di Sale di Camargue oggi è anche IGP!

Leone d’oro a Venezia per Gemma di Mare

Stella Mc Cartney sceglie le Saline della Camargue per il lancio della sua nuova collezione

Compagnia Italiana Sali accelera sulla comunicazione

Siamo passati alla storia!

Il Sale di Sardegna

Il Fior di sale da scoprire - I MEAT

Gemma di mare sostiene A.T.T.A TOSCANA

Un peu de sel? Gemma di mare espone a Parigi

A Porto Viro, nella Casa del Sale
Il nuovo nuovo macchinario per il confezionamento delle bustine di sale iodato da 1 grammo

Sempre più vicino ai consumatori: CIS lancia la sua nuova piattaforma di e-shop

Sulle piste dei Campionati del mondo di sci alpino di Cortina 21.

Il Bicarbonato Gemma di Mare Eletto Prodotto dell’Anno 2021

UNA NOVITÀ PRATICA E… IGIENICA PER LA GDO E L’HO.RE.CA

Novità dell’anno 2020

Mini-campagna sul Corriere della Sera

"Il sale... la nostra storia" in campagna su Youtube

Podio e medaglia di bronzo per il sale viola dell'Himalaya.

CIS dona 2 tonnellate di sale iodato alla Croce Rossa Italiana di Chioggia

CIS INFORMA

Il Kala Namak al SANA 2019

Scopri "Progetto Salute" by Gemma di mare

Compagnia Italiana Sali su La7d

Fornitore ufficiale per MasterChef All Stars Italia

Viviamo insieme la magia del sale... a Cibus